Minor Blood Group Antigens Deleted by Gene Editing
By LabMedica International staff writers
Posted on 13 May 2018
The CRISPR/Cas9 gene-editing tool was used to create a line of red blood cells for transfusion that was completely deficient in blood groups encoded by five different genes that generate antigens responsible for the most common transfusion incompatibilities.Posted on 13 May 2018
Regular blood transfusion is the basis of care for patients with red blood cell (RBC) disorders such as thalassemia or sickle‐cell disease. However, repeated transfusions will often cause patients to develop an immune response to all but the most specifically matched donor blood due to incompatibility at the level of minor blood group antigens.
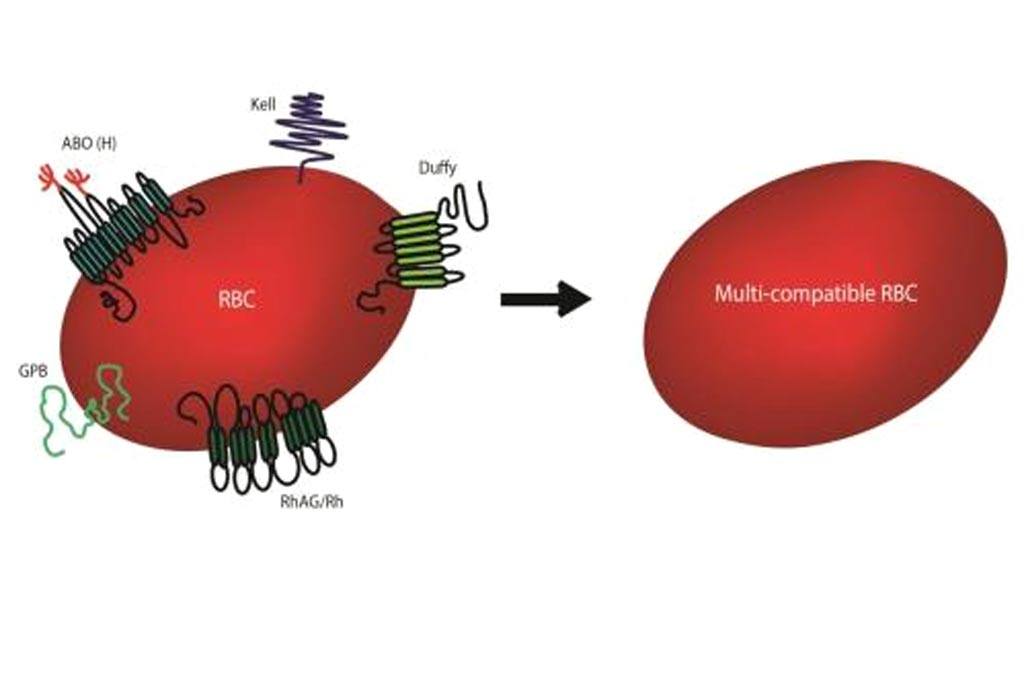
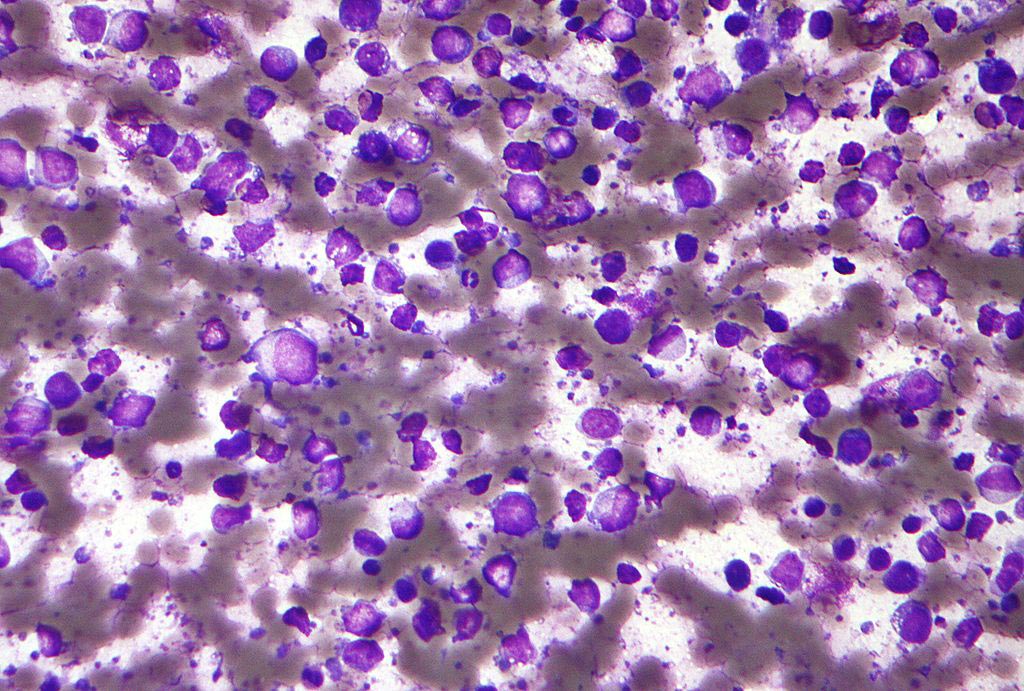
Image: Synthetic biologists have succeeded in generating laboratory-made red blood cells deficient in minor blood group antigens (Photo courtesy of Dr. Ashley Toye, University of Bristol).
To eliminate the most common minor blood group antigens from donor blood cells, investigators at the University of Bristol (United Kingdom) used CRISPR‐mediated genome editing of an immortalized human erythroblast cell line (BEL‐A) to generate multiple competent cell lines deficient in individual blood group antigens.
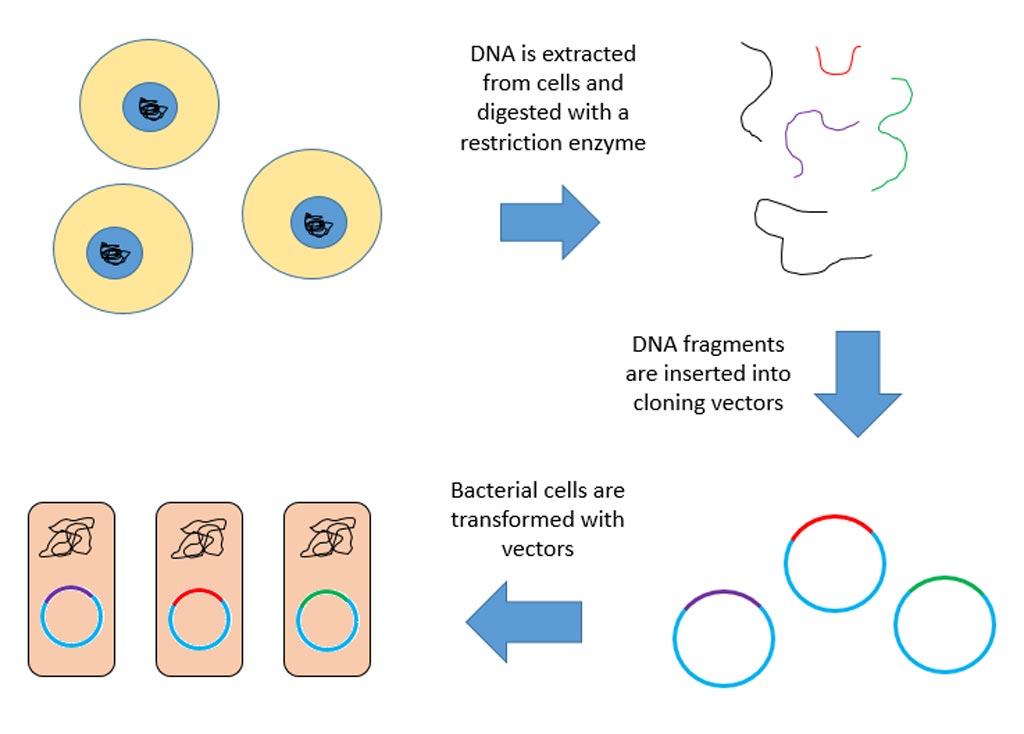
CRISPR/Cas9 is regarded as the cutting edge of molecular biology technology. CRISPRs (clustered regularly interspaced short palindromic repeats) are segments of prokaryotic DNA containing short repetitions of base sequences. Each repetition is followed by short segments of "spacer DNA" from previous exposures to a bacterial virus or plasmid. Since 2013, the CRISPR/Cas9 system has been used in research for gene editing (adding, disrupting, or changing the sequence of specific genes) and gene regulation. By delivering the Cas9 enzyme and appropriate guide RNAs (sgRNAs) into a cell, the organism's genome can be cut at any desired location. The conventional CRISPR/Cas9 system is composed of two parts: the Cas9 enzyme, which cleaves the DNA molecule and specific RNA guides that shepherd the Cas9 protein to the target gene on a DNA strand.
The investigators reported in the April 26, 2018, online edition of the journal EMBO Molecular Medicine that by simultaneously expressing multiple guide RNAs in these cells, they demonstrated the ability to delete multiple blood group genes in erythroblasts and presented proof‐of‐principle generation of red blood cells completely deficient in blood groups encoded by five different genes that encode antigens responsible for the most common transfusion incompatibilities: ABO (Bombay phenotype), Rh (Rh-null), Kell (K0), Duffy (Duffy-null), and GPB (S-s-U-).
Senior author Dr. Ashley Toye, reader in cell biology at the University of Bristol, said, "Blood made using genetically edited cells could one day provide compatible transfusions for a group of patients for whom blood matching is difficult or impossible to achieve within the donor population. However, much more work will still be needed to produce blood cells suitable for patient use."
Related Links:
University of Bristol