Nanoparticles Deliver Small RNAs to Treat Pancreatic Cancer
By LabMedica International staff writers
Posted on 06 Feb 2018
Posted on 06 Feb 2018
A team of Israeli cancer researchers used novel supramolecular nanocarriers to deliver a potent combination of microRNA and siRNA to attach and destroy pancreatic ductal adenocarcinoma (PDAC) tumors in a mouse model.
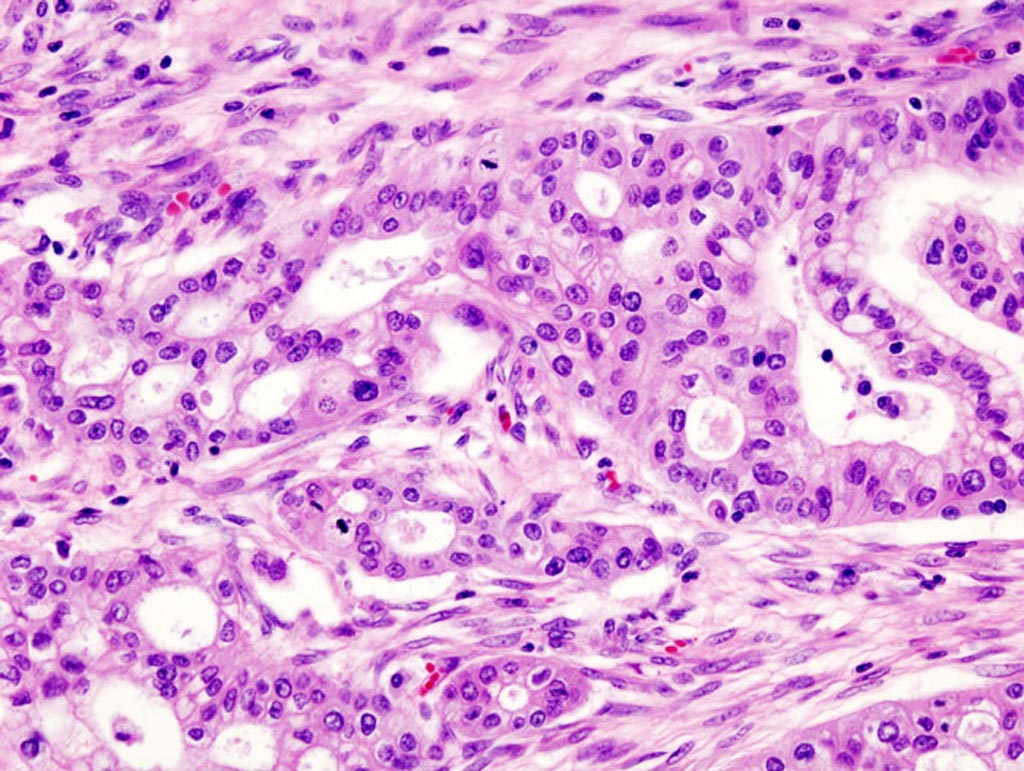
Image: A micrograph of pancreatic ductal adenocarcinoma (the most common type of pancreatic cancer) (Photo courtesy of Wikimedia Commons).
MicroRNAs (miRNAs) are a small noncoding family of 19- to 25-nucleotide RNAs that regulate gene expression by targeting messenger RNAs (mRNAs) in a sequence specific manner, inducing translational repression or mRNA degradation, depending on the degree of complementarity between miRNAs and their targets. Many miRNAs are conserved in sequence between distantly related organisms, suggesting that these molecules participate in essential processes. In fact, miRNAs have been shown to be involved in the regulation of gene expression during development, cell proliferation, apoptosis, glucose metabolism, stress resistance, and cancer.
Short interfering RNAs (siRNAs) have a well-defined structure: a short (usually 21 base pairs) double-stranded RNA (dsRNA) with phosphorylated 5' ends and hydroxylated 3' ends with two overhanging nucleotides. These small RNAs can bind to other specific messenger RNA (mRNA) molecules and either increase or decrease their activity, for example by preventing an mRNA from producing a protein. RNA interference has an important role in defending cells against parasitic nucleotide sequences – viruses and transposons – but also in directing development as well as gene expression in general.
Investigators at Tel Aviv University (Israel) conjugated in parallel ethylenediamine and alkylamine moieties to form positively charged amphiphilic nanocarriers. These cationic nanocarriers were designed to utilize electrostatic-based interactions to form polyplexes with the negatively charged oligonucleotide cargo. The nanocarriers would facilitate oligonucleotide delivery by improving their stability in the bloodstream and enabling accumulation of the polyplexes at the tumor site due to enhanced permeability and the retention effect.
Based findings gleaned from The Cancer Genome Atlas (TCGA), the investigators used two negatively charged small RNAs: miR-34a for miRNA replacement therapy and PLK1 (polo like kinase 1)-siRNA for oncogene silencing in a PDAC mouse model.
They reported in the January 2, 2018, online edition of the journal Nature Communications that systemic administration of the RNA polyplexes to PDAC-bearing mice showed no toxicity and accumulated at the tumor, resulting in an enhanced antitumor effect due to inhibition of the MYC oncogene, a common target of both miR-34a and PLK1.
"Though 75% of pancreatic cancer patients die within 12 months of diagnosis, about 7% survive more than five years," said senior author Dr. Ronit Satchi-Fainaro, professor of physiology and pharmacology at Tel Aviv University. "We thought that if we could understand how some people live several years with this most aggressive disease, we might be able to develop a new therapeutic strategy. This treatment takes into account the entire genomic pattern, and shows that affecting a single gene is not enough for the treatment of pancreatic cancer or any cancer type in general."