Progenitor Cells Show Promise for Repairing Heart Attack Damage
By LabMedica International staff writers
Posted on 21 Apr 2017
Intermediate progenitor cells can be induced to develop into either endothelial cells (the type of cell that lines the walls of blood vessels) or into erythroblasts (the type of cells that mature into red blood cells) depending on the level of the transcription factor SOX17.Posted on 21 Apr 2017
Investigators at the University of Illinois generated CD34+ progenitor cells following the de-differentiation of human adult dermal fibroblasts by overexpression of pluripotency transcription factors. Sorted CD34+ cells were transdifferentiated into induced endothelial cells (iECs) and induced erythroblasts (iErythroblasts) using lineage specific growth factors. The therapeutic potential of the generated cells was assessed in an experimental model of myocardial infarction.
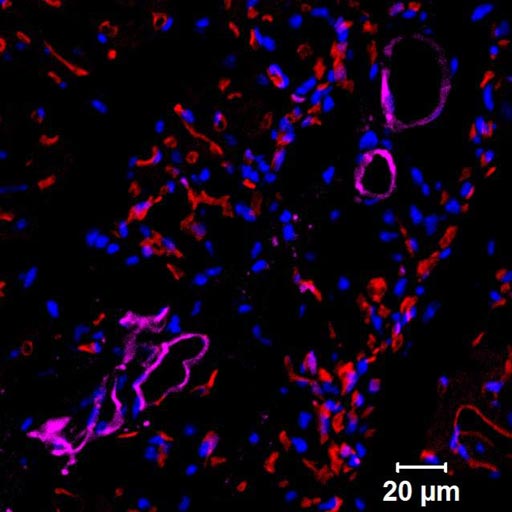
Image: A micrograph of a mouse heart section showing human progenitor cells that formed functional human blood vessels. Purple color signifies human blood vessels, red staining signifies the blood vessels of the mouse that received the human cell implants (Photo courtesy of Dr. Jalees Rehman, University of Illinois).
Results published in the April 5, 2017, online edition of the journal Circulation revealed that iECs expressed specific endothelial cell surface markers and also exhibited the capacity for cell proliferation and neovascularization. Induced erythroblasts expressed erythroid surface markers and formed erythroid colonies. Endothelial lineage conversion was dependent on the upregulation of the developmental transcription factor SOX17 whereas suppression of SOX17 instead directed the cells towards an erythroid fate.
Implantation of these human bi-potential CD34+ progenitors into immune-deficient NOD-SCID mice resulted in the formation of micro-vessels derived from human endothelial cells that were perfused with mouse and human erythrocytes. In addition, iECs generated from human fibroblasts showed upregulation of the enzyme telomerase. Cell implantation markedly improved vascularity and cardiac function after myocardial infarction without any evidence of cancer formation.
Senior author Dr. Jalees Rehman, associate professor of medicine and pharmacology at the University of Illinois, said, "Progenitor cells can be grown in large quantities sufficient for regenerative therapies. And unlike pluripotent stem cells, progenitor cells can only differentiate into a few different cell types. Without understanding the molecular processes, it is difficult for us to control or enhance the process in order to efficiently build new blood vessels."
"The increase in telomerase we see in the progenitor cells could be an added benefit of using this partial de-differentiation technique for the production of new blood vessels for patients with cardiac disease, especially for older patients," said Dr. Rehman. "Their cells may already have shortened telomeres due to their advanced age. The process of converting and expanding these cells in the lab could make them age even further and impair their long-term function. But if the cells have elevated levels of telomerase, the cells are at lower risk of premature aging. We were concerned about the risk of tumor formation. But to truly determine the efficacy and safety of these cells for humans, one needs to study them over even longer time periods in larger animals."